Kill It - The Epstein Files of Science
The Team that Told you Not to Look
[Hello Reader. I’m still here, still documenting, still explaining]
The Research Team Behind the Science: Who Consolidated Candida albicans as an “Opportunistic Pathogen” in 1965
It started, like a lot of this, with a thread I pulled because I was writing and found a curiosity.
ChatGPT mentioned that β-hydroxybutyrate test for ketones was superior to the normal UA or dipsticks. I wondered, “So, why is there more than one test for the same thing?” Well, as it turns out, it isn’t measuring the same thing. The UA tests for acetoacetate in the urine, so it is a delayed reading (post-metabolism, and only if you’re hydrated enough to go). The β-hydroxybutyrate test is a finger prick that directly detects ketones with a fast and precise measurement
So, now, I’m thinking, why doesn’t everyone get that test? Answer: the new one costs more (of course). But, my other question was more related to the when. When was this test developed?
Why did I care? Well, honestly, my mind was a bit different at that point (See Personal Note on the Mental Impact of this Condition In the Last Phase, Redacted Science P 171) The way I tried to describe to people at the time [whom I’m quite sure thought I at least might be crazy] was that that month, while I was writing, I was see things in 3D. When I saw “new test,” I probably thought all those questions at once, not in some sequence. My mind wanted to know everything. My brain saw that it fell right in the sweet spot for the second cohort, or at least when they would be needing such tests to try and manage a possible second cohort. If you are running a classified Bourne Identity-type project, you need to know exact ketone levels right now, not approximate ones that reflect what your body was doing sometime recently. Otherwise, yeah, it’s helpful, but diabetes wasn’t a huge thing yet. Sure, it would help diabetics, which made it “reasonable” to be studying and “releasable” (ie, profitable) to science, even though the real reason you might be doing it was a study breaking the Nuremberg Code [These are fundamental principles of centralized institutional medicine. Clickety click for more]
So I started looking at who developed this cool new test, when, and where. I pulled on the first one, D.H. Williamson. Where was he? What was he studying? Who else was at or even near that place, and what were they studying that had to do with anything I was writing about? Not “did they collaborate.” That was immediately discounted. Why? See above. No one breaking that code is going to publish together in diverse areas. They might as well shine a spotlight on themselves.
The answer to my question was a cluster of names publishing within months of each other in the early 1960s, all working on fuel prioritization and metabolic switching: HA Krebs 1(1953 Nobel Prize for identifying the Krebs cycle – how cells generate ATP) [Boom, that was a word that exploded in my head. You see, the Article said “this condition is all about ATP “ – Redacted Science P62], P.J. Randle, Williamson, Peters. I also had Mellanby and Earl Sutherland Jr. on my first-pass list, too. Not bad for working from memory and ChatGPT at 2 AM, but not right either. Sutherland was at Vanderbilt, not in the UK corridor. Mellanby’s connection was thinner than I thought. The names that survived the audit were tighter and more interesting.
But even just the ones I had right were like the ’95 Chicago Bulls, or the ’85 Chicago Bears. This team was made of all-stars. All in one place at the same time. [Things that make you go hhmmm?]
Yes, that’s more like the 2025 Thunder and the 95 Bull’s. Look at that list.
D.H. Williamson was at the Metabolic Research Laboratory, Radcliffe Infirmary, Oxford, working directly in Krebs’s group. He co-published the early β-hydroxybutyrate work that nailed down the metabolic switches I was reading about. What I didn’t know then was where Williamson went next: lactation biochemistry. The biochemistry of milk production. If I had known? I wouldn’t have care. It wouldn’t have clicked yet. I had not done the research on c. albicans at the level I have now. Yes, I had figured out it was a biochemical computer, but I hadn’t figured out the ecological staging environment. That pivot matters enormously if you understand that lactation is the primary transmission architecture for C. albicans colonization of the infant gut.
Williamson’s published focus was on metabolic regulation. Lactation is the one phase where the host voluntarily runs a version of the program candida can run inside the host. The body suppresses its own needs to feed another organism through triggering insulin resistance, sparing the glucose, redirecting lipids, and prioritizing the recipient. He traced the metabolic substrate pathway that leads directly to the organism’s ecological establishment in every new human being.
He had the cleanest model system in mammalian biochemistry sitting in front of him. Lactation as a metabolic extreme: tissue-specific insulin resistance, substrate rerouting, fuel prioritization, the whole thing switching on and off with litter removal. And he never once looked at what was being transmitted to the pups through the milk. He may not have known that’s what he was mapping, but that’s what he mapped. [If he DID know, that might mean there is a lot of interesting lactation research out there that never got published. I wonder why not? Oh, yeah, the Redacted Science part. If they let you know you have an evolutionary companion symbiont, that might create waves. Real waves, financial waves, institutional waves, religious waves, societal waves, Fourth Turning level waves with evolutionary existentialism thrown in. Oh, but it’s FINE…they redacted it. That fixed everything, right? No one is EVER going to figure out because we’ll tell them c. Albicans is bad. Kill it. It only shows up if someone is dying. That way, they will never think to look at it. Waves!]
R.A. Peters preceded Krebs in the Oxford biochemistry chair. His fluoroacetate work defined substrate-level toxicology, the idea that you could poison a metabolic pathway by inserting the wrong molecule at the right step. That’s the intellectual framework that makes the organism’s arachidonic acid competition comprehensible: C. albicans doesn’t poison the pathway. It competes for the fuel source at the branch point between prostaglandin and endocannabinoid synthesis. Think of having your hand on two knobs, one labeled inflammation and one labeled the body’s own response to it. [No, actually stop and think about that for at least 10 seconds, it is a bit scary]. Peters would have understood this immediately. He was retired by the time it mattered, but he and his thinking were still in the walls – Professor Emeritus, technically, but I would later find his name actively involved.
I.M. Glynn was at the Cambridge Physiological Laboratory, characterizing the Na⁺/K⁺-ATPase with Garrahan. Their 1967 paper demonstrated that the sodium-potassium pump could run in reverse, harvesting energy from ion gradients instead of consuming ATP to build them. That paper is the origin of the pump reversal anecdote I use in Paper B: the lead internal medicine faculty member, unaware that the pump could reverse, despite Garrahan and Glynn having published it nearly 60 years earlier. The data exists. The attention doesn’t. [I will spare you about how I was lectured by another senior Internal Medicine physician about how the Na⁺/K⁺ couldn’t go in reverse. I mean, I can forgive my PCP, but these guys should know their stuff]
P.J. Randle, Cambridge and Bristol, proposed the glucose-fatty acid cycle in 1963. The Randle cycle is the mechanistic explanation for metabolic switching between carbohydrate and fat oxidation at the cellular level. This happens inside a cell when both fuel sources are present. If the cell starts burning fat, the byproduct (Acetyl-CoA) physically blocks the enzymes needed to burn glucose. It’s a “this or that” mechanism for cells that can use both. If you understand the Randle cycle, you understand insulin resistance as a fuel-competition event rather than a receptor defect. That understanding is central to everything in my framework. What’s being burned when and why and how.
The Smoking Gun [or annual report]
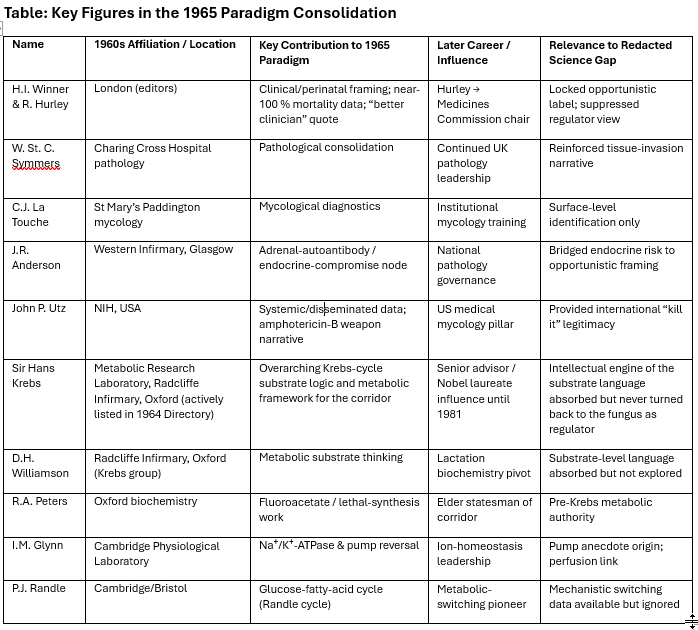
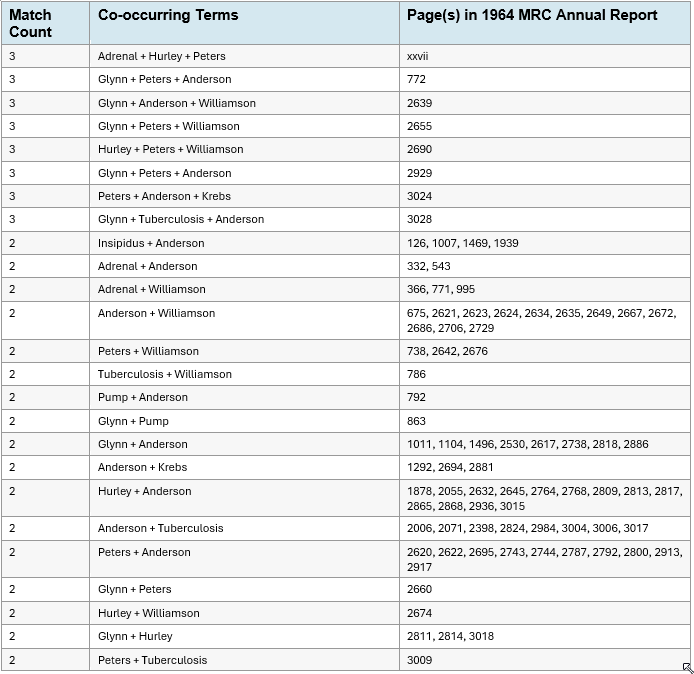
The 1964 Medical Research Council Annual Report and Handbook — the official directory of MRC-funded programs, committee memberships, and grant allocations for that year — contains page-level co-occurrences among researchers who never co-authored a publication [it’s tricky, you cannot read it online, but you can search one word terms, compiling this list took a bit of work]. A systematic keyword search of this document reveals that Rosalinde Hurley (perinatal Candida, Charing Cross), R.A. Peters (metabolic toxicology, Oxford), D.H. Williamson (mitochondrial metabolism, Oxford), I.M. Glynn (Na⁺/K⁺-ATPase, Cambridge), J.R. Anderson (adrenal autoimmunity, Glasgow), and Hans Krebs (Nobel laureate, metabolic biochemistry, Oxford) appear together repeatedly across committee rosters, program summaries, and grant sections. Hurley and Peters share front-matter page xxvii alongside “adrenal.” Glynn, tuberculosis, and Anderson converge on page 3028. Peters, Anderson, and Krebs appear together on page 3024. Anderson is the sole named researcher co-occurring with “insipidus” on four separate pages. These are researchers in different disciplines, at different institutions, who published in different journals and attended different conferences. None of them co-authored work that crossed the boundary between mycology, metabolic biochemistry, and immunopathology. Yet the MRC’s own records place them in the same governance and funding sections, one year before Winner and Hurley convened the symposium that defined Candida albicans as an opportunistic pathogen and excluded every metabolic and biochemical question from the sanctioned conversation. The published literature kept these programs apart. The MRC’s internal paperwork shows they were together. These are areas that overlap precisely in the framework of Redacted Science, but otherwise, they are quite difficult to explain.
MRC 1964 Annual Report — Cross-Researcher Page Co-occurrences
Keyword searches performed on the 1964 Medical Research Council Annual Report and Handbook. Each row represents a unique combination of researcher names or medical terms appearing on the same page. “Anderson” refers to J.R. Anderson (adrenal autoimmunity, Glasgow). Pages grouped by term combination.
Figure 2
Note: These researchers worked in different disciplines (mycology, metabolic biochemistry, immunopathology, membrane physiology), at different institutions (London, Oxford, Cambridge, Glasgow), and did not co-author publications crossing these disciplinary boundaries. Their co-occurrence in MRC governance records documents institutional connectivity that the published literature does not reveal.
These men weren’t mycologists. They were biochemists. But their grant-panel memberships, committee overlaps, and MRC directory listings placed them on the same pages as the clinical team that was about to define Candida albicans for the next sixty years. They supplied the mechanistic language (perfusion, electrolyte balance, metabolic switching) that the clinical narrative absorbed without ever asking whether the organism might be running those programs deliberately.
They said “kill it” so that people wouldn’t look closer. Let’s think about that one, too. Science and progress only accelerate, folks. They knew they were kicking the can down the road. How far? I have no idea, but I bet they would be shocked if you told them back then that they would buy 60 years with that approach.
At this point, the people who made the decision are long gone. The people holding onto the knowledge? I have no idea.
Figure 3 The First Ever Symposium on Candida Infections (1965)
The clinical team was compact. In May 1965, at the Royal College of Physicians in London, H.I. Winner and Rosalinde Hurley convened the first-ever Symposium on Candida Infections. The proceedings were published the following year by E. & S. Livingstone. The publication numbers tell the story: annual publications on candidiasis had climbed steadily from 67 in 1959 to 111 in 1963. Winner and Hurley published their monograph in 1964. The next year, publications dropped 19%, to 87. Then the symposium rebooted the field. By 1966 the count was 129 and climbing. 150 in 1967. 191 in 1970. 235 in 1971. The curve never came back down. But it grew within the framework the symposium had established. The questions that got asked after 1965 were the questions Winner and Hurley had sanctioned.
W. St. C. Symmers chaired the symposium from Charing Cross Hospital pathology, wrote the introduction, and presented on septicaemic candidosis. He framed the narrative: Candida was a minor pathogen that modern medicine (antibiotics, steroids, cytotoxic drugs) had accidentally unleashed. C.J. La Touche brought the mycological credentials from the University of Leeds. La Touche is the mycologist whose mould contaminated Fleming’s plate and gave the world penicillin. The man who accidentally created the antibiotic era was now chairing the symposium on the fungal infections that era had amplified. That’s not irony. That’s a closed loop. A neatly-tied bow around it all.
J.R. Anderson sat at the Western Infirmary, Glasgow. His MRCPath in 1964, his adrenal-autoantibody work with Goudie and Gray, mapped the endocrine-compromise lane that overlapped with opportunistic candidiasis. Addison’s disease (adrenal insufficiency) appears in Winner’s own tables as a primary condition associated with Candida infection. Anderson bridged endocrine vulnerability to the opportunistic framing. His later rise to national pathology governance gave the Glasgow node institutional weight that outlasted the symposium by decades.
The lone American was John P. Utz of the NIH. Utz supplied systemic candidosis data from leukemia patients and the explicit endorsement of amphotericin B as the weapon. His chapter completed the circle: Candida was an aggressive killer in compromised hosts, and the response was pharmaceutical.
Now, everyone was on the same page – opportunistic infection, kill it, look for what’s damaged afterwards. If you read the The Saline Oscillation Hypothesis: Endocannabinoid-Mediated Fungal-Hominid Co-Evolution in the East African Rift Valley [You get paid by the word in the title I swear] you’ll understand what that really means. But, let’s just say it’s probably costing lives one way or the other. [Oh, that isn’t quite out yet, as of this writing, but it is nearly done…subscribe to be among the first!]
Hurley’s data on untreated systemic candidosis showed mortality approaching 100%. Winner’s memorable observation that “Candida is a better clinician than the doctor” acknowledged that the organism detected disease states physicians had missed. Read in 1965, that line reinforced the pathogen narrative. Read in 2026, it sounds like a description of a distributed sensing system calibrated to host physiology. But, not to a silo’ed physician who knew before they were going to med school they would end up just studying part of it, and later find out they were really just following decisions trees someone else built.
The collective message was unambiguous: Candida albicans is an opportunistic pathogen. Kill it. The “opportunistic” label became the dominant paradigm. [Trust me when I say doctors have explicit one-way view on candida. This is drilled into them, and it is a check-valve on logic.] But the organism’s behavior under stress (rapid morphological switching, parasexual recombination, morphology-dependent extracellular vesicle cargo, bet-hedging across micro-niches) is precisely what a distributed biochemical computer deploys when confronting unknown circumstances. What the 1965 team read as exploitation was adaptive computation.
Krebs and Sir George Alberti sit at the junction of this network as the senior institutional figures whose training and access are consistent with the cold-eyed metabolic observations I read in 1995 about the first early 20th century cohort. Alberti was Krebs-trained at Oxford, DPhil completed in 1964 (the same year as Winner and Hurley’s monograph), with a career built on diabetic ketoacidosis and insulin pharmacology. Now Senior Research Investigator at Imperial College London, the institution formed by merging St Mary’s (La Touche), Charing Cross (Winner, Hurley, Symmers), and the Royal Postgraduate Medical School. The metabolic corridor and the clinical corridor converged into his institutional address.
The net is small but professionally interlocked. Geographically dispersed (Glasgow, London hospitals, Oxford, Cambridge) yet bound by MRC grant listings, Medical Directory pages, and national committee memberships, with one NIH voice supplying transatlantic legitimacy. In a single year, this ecosystem institutionalized the “kill it” paradigm across Anglo-American clinical literature. The deeper co-evolutionary reality remained invisible.
They didn’t know an article would come out later, and also get redacted. That article was enough for someone that saw it [ahem…] to replicate the original experiment and go on to document it all
The cost is now measurable. Despite contributing substantially to global infectious disease mortality, fungal pathogens historically received on the order of 1-2 % of infectious-disease research funding, reflecting a systemic neglect of fungal ecological systems within modern biomedical science.
Sixty years after the 1965 symposium, the World Health Organization published its first fungal priority pathogens list. 2022. Not a data gap. An attention gap, operating at every layer from bench funding to medical education to the clinical encounter where a doctor has never heard that the sodium-potassium pump can run in reverse. They decided to ignore it. The longitudinal human dataset that would have forced different questions was removed from the record. Six decades of integrated inquiry in microbiology, immunology, metabolism, and host-microbe ecology, lost. The phenotype that may have helped build early civilization was erased from the scientific canon. And the organism kept computing.
What have we not learned? We cannot possibly know. But, I’m getting us back on track. Sure, we’re 60 years late and it is a Fourth Turning, but they won’t redact it again. Terms like Homo Candidus will step into science and new discoveries will be made.